Specimen Collection Manual: Tissue (Skin, Sub-Cutaneous, Etc.), Fluid Collection - Viral
Instructions
Materials/Supplies
- Sterile syringe, 2-5 mL.
- Sterile screw-capped collection container.
- UTM or UVT transport medium.
Specimen Collection Criteria
- Tissues: Collect tissues aseptically taking care to prevent cross-contamination when specimens are taken from multiple sites.
- Use different tubes of transport media for tissue specimens from different sites.
- Autopsy specimens should be collected within 24 hours of the time of death
- Vesicle Fluid Collection: Select a vesicle containing clear fluids. The efficiency of virus detection decreases significantly when pustular fluids are collected.
- Aseptically unroof the lesion and blot the excess fluid with a swab.
- Gently rub the base of the lesion with a flocked ESwab to collect infected epithelial cells.
- Place the swab in viral transport medium and break the shaft at the perforation so that it will fit into the vial.
- Fluids: Aspirate into a sterile syringe (2-5 mL). Transfer the aspirate into a sterile, screw-capped collection container.
Rejection Criteria
- Do not submit tissues for culture in formalin, or in containers that ever contained formalin.
- Do not use expired media. Check expiration date on all media prior to use.
In-Lab Processing
- Small tissue specimens should be placed into viral transport medium.
|
If the Specimen is for:
|
Then:
|
|
Viral Culture
|
Small tissue specimens should be placed into UTM or UVT transport medium.
|
|
Fluids
|
Do NOT place into UTM or UVT. Submit in a sterile, screw-capped collection container.
|
- Send specimens to the Laboratory on wet ice or with a cold pack. Do not freeze. Specimens may be stored refrigerated (2-8°C or 36-46°F) for up to 24 hours. If longer delays are anticipated, specimens should be frozen at -70°C.
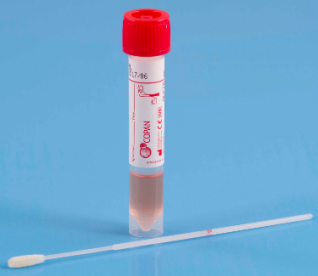 |
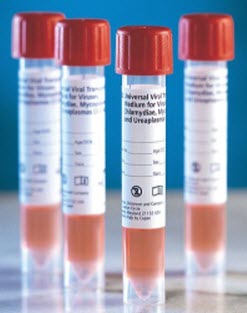 |
| UTM |
UVT |
Contacts
Microbiology Laboratory – RO
248-551-8090
Name: Microbiology Laboratory – RO
Location:
Phone: 248-551-8090
Microbiology Laboratory – TR
248-964-6127
Name: Microbiology Laboratory – TR
Location:
Phone: 248-964-6127
Microbiology Laboratory – GP
313-473-1802
Name: Microbiology Laboratory – GP
Location:
Phone: 313-473-1802
Last Updated
1/7/2020
Microtainer® and Vacutainer® are registered trademarks of Becton, Dickinson and Company.
UroVysion® is a registered trademark of Abbott Laboratories. ThinPrep® is a registered trademark of Hologic, Incorporated.